Unit 3 - The Particle Theory of Matter
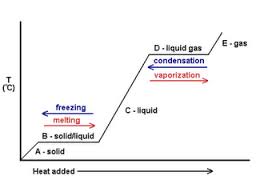
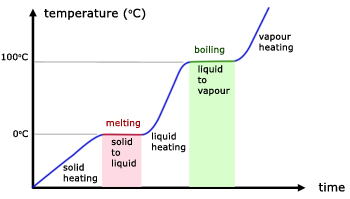
Boiling Point ------- The temperature at which a liquid begins to boil and change into a gas or vapour.
Ex: Water is a substance with a boiling point of 100 degrees Celsius (C).
Celsius (C) Scale ----- The most common scale for measuring temperature (used in Canada).
Chemical Change ------ A change in in matter (usually due to a mixture) that causes at least one new substance, with new properties, to be
formed. In a chemical reaction molecules are broken and reformed in a new way.
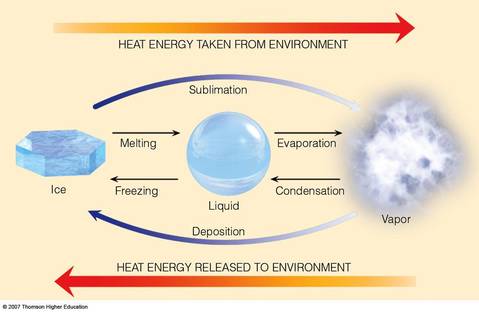
Condensation --------- The process of changing from a gas (or vapour) to a liquid.
Ex: Clouds, fog, and dew are examples of condensation
Condensation Point ------ The temperature at which a gas or vapour begins to change into a liquid
Conduction -------- This is a method of Heat Transfer occurring in solids (or from solid to solid). Thermal energy directly transfers from
one particle or object to another through contact.
Convection -------- This is a method of Heat Transfer occurring within liquids and gases. Convection creates currents
(continuous circulation of a liquid or a gas when heat is transferred from hotter areas to colder areas)
Contract ------- When matter contracts, the particles get closer together (looks like shrinking or tightening). When energy (heat) is
taken away from a substance rapidly it may contract
Density ---------- Density is a measure of how solid (closely packed) particles are in a given substance. The more dense, the more tightly
packed the particles, and the heavier it would be.
Dilute (Diluted) ------ To weaken the strength of a solution by increasing the amount of solvent
Dilute Solution ------- A solution that contains relatively little solute
Dissolving -------- Mixing a solute completely with a solvent to form a solution; the distinct properties of each of the materials combine
into one set of properties (homogeneous). The particles of each substance evenly distribute throughout the mixture.
Distillation ------- A process for separating the parts of liquid solution (ex: saltwater). The solvent is heated to change into a gas and it
will evaporate... the solute particles are left behind. The gas is then collected and condensed back into a liquid.
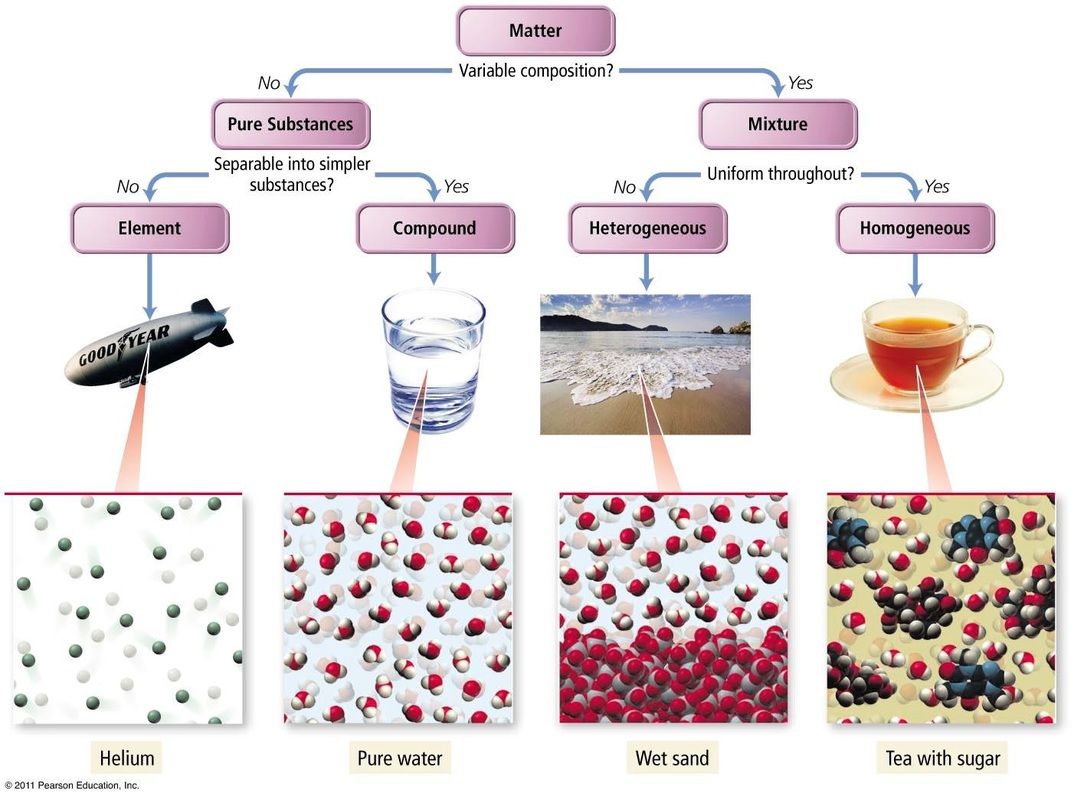
Element -------- A type of pure substance (made from one specific type of atom) that cannot be broken down into smaller parts. Each atom
has a unique set of properties (characteristics).
Evaporation -------- The process by which a liquid, such as water, changes into a gas or vapour.
Expand --------- When matter expands, the particles move farther apart and the volume increases. When energy (heat) is added to a
substance rapidly it will give energy to the particles which will spread out and move at a more rapid rate.
Gas --------- A state (phase) of matter. A gas has no particular shape or size and can be compressed. The particles in a gas have the
highest energy and motion (they move rapidly and randomly).
Heat ------------ Thermal Energy transferred from one object or substance to another. Temperature is the way HEAT is MEASURED.
Heat Insulators ------ Materials that slow the transfer or conduction of heat energy from one place to another.
Heterogeneous Mixture ----- A mixture made up of parts that retain their own properties. You can visibly see more than one type of matter.
Homogeneous Mixture ----- A mixture that blends together so that only one set of properties can be identified
(looks consistent throughout).
Insoluble -------- A substance that is not able to dissolve another substance (a solute that cannot dissolve into a solvent).
Ex: Oil and Water are insoluble. They will not mix to become a solution (homogeneous)
Luster ------- A property of matter (characteristic) that allows light to reflect and create a sheen or shine.
Matter ------- Matter is anything that takes up space (has volume), has a mass, and is made up of particles.
Mechanical Separation ---- picking things out with tongs, separating parts of a mixture by hand (ex - separating smarties by colour)
Melting ------ A phase change where a solid changes to a liquid
Melting Point ----- The temperature at which solid matter begins to change to a liquid
Properties ------- The characteristics (traits) of matter in different materials and substances. Every substance has its own unique set of
properties; examples of properties include colour, odour, density, texture, luster, conductivity, malleability...etc.
Pure Substance ------ A material that is composed (made up of) only one type of particle (atom or molecule). Examples of pure substances
include gold, oxygen, and water (if the water has been purified or distilled).
Radiation --------- The transfer of energy in the form of waves. Radiant heat can travel through space and do not need to travel through any
type of matter (solid, liquid, or gas) but are able to move through empty space. The sun is a great example of radiation...
the suns rays will travel through space to reach the earth.
Solution ---------- A solution is a mixture that occurs when one substance dissolves into another creating a homogeneous mixture. The
mixture blends together looking like one set of properties... when in fact... there are two or more sets of properties.
Saturated Solution ------- A saturated solution can occur when too much solute is added to a solvent and the spaces between particles become
full. The solvent will no longer be able to dissolve the solute particles. When there are no spaces left for dissolved
particles this is called saturation. Any new particles added will sink to the bottom and not mix into the solution.
Soluble -------- A substance that is able to dissolve into a solvent (solute) is soluble. If two things are soluble it means their mixture will
create a solution or homogeneous mixture.
Solute --------- A substance that can be dissolved in a solvent. Ex: Salt or Sugar would be a solute (can be dissolved in water)
Solvent --------- A substance that is able to dissolve other substances. For example water is known to be a universal solvent
(able to dissolve many things such as salt, sugar, minerals, and other chemicals)
Sublimation ------- The process in which a solid changes directly into a gas (skipping the liquid phase).
Temperature ------ Temperature is the measure of the amount of heat within a substance or object.
Theory ---------- A theory is an explanation of an event that has been supported by consistent, repeated, experimental results and has
therefore been accepted by many scientists. As new technology and discoveries are made theories may change!
Thermometer ------- A device used to measure temperature
States of Matter
Solid - a state of matter with a fixed (definite) shape and volume. The particles in a solid have low energy, do not move much only
vibrate, and the particles are highly attracted to one another.
Liquid - a state of matter with a fixed (definite) volume but an INDEFINITE shape (usually will take the shape of the container). The
particles in a liquid have medium energy and are able to flow and move past one another.
Gas - a state of matter with an indefinite (undefined) shape and volume. The particles in a gas are in rapid random motion, have high
energy,and are spaced far apart (low attraction between particles).
Heating Curve (Phase Changes shown in a graph)
Ex: Water is a substance with a boiling point of 100 degrees Celsius (C).
Celsius (C) Scale ----- The most common scale for measuring temperature (used in Canada).
Chemical Change ------ A change in in matter (usually due to a mixture) that causes at least one new substance, with new properties, to be
formed. In a chemical reaction molecules are broken and reformed in a new way.
Condensation --------- The process of changing from a gas (or vapour) to a liquid.
Ex: Clouds, fog, and dew are examples of condensation
Condensation Point ------ The temperature at which a gas or vapour begins to change into a liquid
Conduction -------- This is a method of Heat Transfer occurring in solids (or from solid to solid). Thermal energy directly transfers from
one particle or object to another through contact.
Convection -------- This is a method of Heat Transfer occurring within liquids and gases. Convection creates currents
(continuous circulation of a liquid or a gas when heat is transferred from hotter areas to colder areas)
Contract ------- When matter contracts, the particles get closer together (looks like shrinking or tightening). When energy (heat) is
taken away from a substance rapidly it may contract
Density ---------- Density is a measure of how solid (closely packed) particles are in a given substance. The more dense, the more tightly
packed the particles, and the heavier it would be.
Dilute (Diluted) ------ To weaken the strength of a solution by increasing the amount of solvent
Dilute Solution ------- A solution that contains relatively little solute
Dissolving -------- Mixing a solute completely with a solvent to form a solution; the distinct properties of each of the materials combine
into one set of properties (homogeneous). The particles of each substance evenly distribute throughout the mixture.
Distillation ------- A process for separating the parts of liquid solution (ex: saltwater). The solvent is heated to change into a gas and it
will evaporate... the solute particles are left behind. The gas is then collected and condensed back into a liquid.
Element -------- A type of pure substance (made from one specific type of atom) that cannot be broken down into smaller parts. Each atom
has a unique set of properties (characteristics).
Evaporation -------- The process by which a liquid, such as water, changes into a gas or vapour.
Expand --------- When matter expands, the particles move farther apart and the volume increases. When energy (heat) is added to a
substance rapidly it will give energy to the particles which will spread out and move at a more rapid rate.
Gas --------- A state (phase) of matter. A gas has no particular shape or size and can be compressed. The particles in a gas have the
highest energy and motion (they move rapidly and randomly).
Heat ------------ Thermal Energy transferred from one object or substance to another. Temperature is the way HEAT is MEASURED.
Heat Insulators ------ Materials that slow the transfer or conduction of heat energy from one place to another.
Heterogeneous Mixture ----- A mixture made up of parts that retain their own properties. You can visibly see more than one type of matter.
Homogeneous Mixture ----- A mixture that blends together so that only one set of properties can be identified
(looks consistent throughout).
Insoluble -------- A substance that is not able to dissolve another substance (a solute that cannot dissolve into a solvent).
Ex: Oil and Water are insoluble. They will not mix to become a solution (homogeneous)
Luster ------- A property of matter (characteristic) that allows light to reflect and create a sheen or shine.
Matter ------- Matter is anything that takes up space (has volume), has a mass, and is made up of particles.
Mechanical Separation ---- picking things out with tongs, separating parts of a mixture by hand (ex - separating smarties by colour)
Melting ------ A phase change where a solid changes to a liquid
Melting Point ----- The temperature at which solid matter begins to change to a liquid
Properties ------- The characteristics (traits) of matter in different materials and substances. Every substance has its own unique set of
properties; examples of properties include colour, odour, density, texture, luster, conductivity, malleability...etc.
Pure Substance ------ A material that is composed (made up of) only one type of particle (atom or molecule). Examples of pure substances
include gold, oxygen, and water (if the water has been purified or distilled).
Radiation --------- The transfer of energy in the form of waves. Radiant heat can travel through space and do not need to travel through any
type of matter (solid, liquid, or gas) but are able to move through empty space. The sun is a great example of radiation...
the suns rays will travel through space to reach the earth.
Solution ---------- A solution is a mixture that occurs when one substance dissolves into another creating a homogeneous mixture. The
mixture blends together looking like one set of properties... when in fact... there are two or more sets of properties.
Saturated Solution ------- A saturated solution can occur when too much solute is added to a solvent and the spaces between particles become
full. The solvent will no longer be able to dissolve the solute particles. When there are no spaces left for dissolved
particles this is called saturation. Any new particles added will sink to the bottom and not mix into the solution.
Soluble -------- A substance that is able to dissolve into a solvent (solute) is soluble. If two things are soluble it means their mixture will
create a solution or homogeneous mixture.
Solute --------- A substance that can be dissolved in a solvent. Ex: Salt or Sugar would be a solute (can be dissolved in water)
Solvent --------- A substance that is able to dissolve other substances. For example water is known to be a universal solvent
(able to dissolve many things such as salt, sugar, minerals, and other chemicals)
Sublimation ------- The process in which a solid changes directly into a gas (skipping the liquid phase).
Temperature ------ Temperature is the measure of the amount of heat within a substance or object.
Theory ---------- A theory is an explanation of an event that has been supported by consistent, repeated, experimental results and has
therefore been accepted by many scientists. As new technology and discoveries are made theories may change!
Thermometer ------- A device used to measure temperature
States of Matter
Solid - a state of matter with a fixed (definite) shape and volume. The particles in a solid have low energy, do not move much only
vibrate, and the particles are highly attracted to one another.
Liquid - a state of matter with a fixed (definite) volume but an INDEFINITE shape (usually will take the shape of the container). The
particles in a liquid have medium energy and are able to flow and move past one another.
Gas - a state of matter with an indefinite (undefined) shape and volume. The particles in a gas are in rapid random motion, have high
energy,and are spaced far apart (low attraction between particles).
Heating Curve (Phase Changes shown in a graph)
Phase Changes
States of Matter - Particles
Matter - Pure Substances - Mixtures
Unit 2 - Forces and structures
Force ------ A push or a pull that acts on an object. Force can cause motion unless another (equal) force is acting in the
opposite direction. Force can be shown as a vector (arrow) in a picture.
Force can require contact (two objects touching each other). Ex: Picking Up/throwing a ball (contact between hand and ball)
Some force does not require contact... gravity and magnetism act without contact only proximity (objects coming close together).
External Force ------- A force that acts on the outside of an objects material. (Outside does not necessarily mean outdoors... you can be
inside of a building... but outside of the materials). (Examples: Wind, Snow, Rain, Weight (of objects or the structure itself).
Dead Load: The external force of the WEIGHT of the Structure on itself.
Live Load: The external force of the WEIGHT of the objects and things inside or on a structure, as well as the other external
forces such as the wind, snow...etc.
Internal Force ------ A force that acts on the inside of the material of an object or structure. (Tension, Compression, Shear, Torsion)
Compression ----- A squeezing force (crushing, compacting).
Ex: Lower columns of a skyscraper are compressed by the heavy weight above them
Ex: Footprints compress the soil/ground
Ex: Garbage truck compresses the garbage to take up less space
Tension ------- A pulling force that stretches a material.
Ex: Tug-of-War with a rope
Ex: Rock Climbing (the rope in tension holding the load of a person
Ex: The cables on a suspension bridge holding up the dead load and live load.
Torsion ------- A twisting force that "turns" a material in two opposite directions.
Ex: Twisting a twizzler (liquorice)
Ex: Twisting skin on your arm (Snake-Bite)
Ex: Wringing out a wet towel
Shear ------- Bending or tearing a material due to two forces offset in opposite directions. Shear is the result of
two forces acting in opposite directions sliding past each other.
Ex: Using Scissors (sometimes called "shears"
Ex: Earth/Roadways sliding past each other and breaking due to shear force caused by an EARTHQUAKE.
Structure -------- A structure must have a definite shape/size and function (purpose).
Structures can be NATURAL (made by nature) or MANUFACTURED (man-made... influenced and made by people).
Centre of Gravity ------ A specific point where all of the mass of the structure is evenly distributed around. Gravity acts on all parts
of the structure and it will be stable if all parts are spread equally around the centre of gravity.
4 Principles of Stability: 1) Build a firm foundation
2) Balance Forces
3) Keep thrust lines vertical (Centre of gravity)
4) Use rapid rotation
Solid Structure (also called Mass Structure) -------- A structure made by the piling up (stacking) of materials
(Ex: Pillars, Snow Forts, Pyramids, Brick Walls, Mountains)
Shell Structure -------- A type of structure that obtains its strength from a thin, carefully shaped outer layer of material and
that requires no internal frame. Shell structures are strong and hollow and can support loads.
(Ex: Igloo, Egg-Shell, Cardboard Box, Garbage Bin, Wasps Nest)
Frame Structure -------- A type of structure in which a skeleton of materials supports the weight of the other parts. Frames
require different components to come together and rely on each other for support.
A joint is a point (such as a corner) where two pieces of the frame or structure are fastened together.
Stress (Structural Stress) ------- Structural stress occurs when a combination of external and internal forces act on a structure one at a
time causing changes. Stress is normal in any structure. It does not necessarily mean any changes are permanent... they may
be temporary changes. Example: When kicking a soccer ball there is a momentary compression within the material of the ball
when the foot contacts the ball.
Fatigue (Structural Fatigue) ------- Damage due to a high level of structural stress over time. Example: If a large amount of pressure is
placed on an egg there will be some compression within the shell, some tension on the surface, and these forces may lead to
cracking the shell. The cracks represent structural fatigue (damage).
Failure (Structural Failure) ------- Stress can lead to fatigue which can become failure with increased force on a structure. When a structure
FAILS it is no longer able to serve its purpose/function. It will also no longer maintain its shape, size, and form.
Structural Efficiency ------- A single number that compares the mass of a structure with the load it supports.
A high structural efficiency is a good thing.
Structural Efficiency = Maximum Mass Supported by the Structure
Mass of the Structure (Dead Load)
Foundation ------ A solid base of a structure. A firm foundation is important for stability.
Ex: The foundation of a modern home here in Winnipeg would consist of a few layers of concrete. A foundation can
prevent the heavy dead load of a structure from sinking into the ground.
Ex: Feet are generally the "foundation" or base of a human structure. The most stable position is to have both feet firmly
planted on a solid surface. Standing on tip-toes reduces the surface area on the ground and becomes an unstable foundation.
Arch ------ A strong shape often used in structural design. An arch can spread force evenly through the whole structure and down to
the ground. Ex: The Catenary Arch forms were used in the Ice Hotel Design. The arch passageways were able to evenly
distribute the weight of the snow and ice piled above it.
Corrugates ------ Corrugates or corrugation helps resist compression within a material by creating a series of arches inside of the
material. The most common example of this is corrugated CARDBOARD boxes. Corrugation increases strength and
stability of a material by using shape!
Braces, Ties, Gussets ------ A brace, tie, and gusset form a triangle in the corner of a frame or joint (where two pieces are brought
together). They change rectangular and square shapes into triangles for additional strength and stability.
Guy Wire -------- A wire used to give stability to a structure usually by counter-balancing potentially dangerous forces.
Truss -------- A frame structure that uses triangles for strength. (Ex: A Truss Bridge)
Mass -------- The mass of an object is the measure of the amount of material in it. Measurements of mass tell something about the tiniest
particles in the object.
Weight --------- Weight is similar to mass, however, it also includes the force of gravity. On the moon, you would have the same mass,
however you would weigh 1/6 less on the moon because the force of gravity is not as strong.
Gravity --------- In the 17th Century Isaac Newton realized there is a force between any two objects, anywhere in the universe, that tries to
pull them together. Newton analyzed the size of this force, which he called gravity, depended on the mass of the two objects. The
gravitational force between two very small objects is extremely small. You do not notice gravity pulling you toward trees or other
people but the force is there. Earth however, is big enough for the gravitational force between it and nearby objects to be important.
opposite direction. Force can be shown as a vector (arrow) in a picture.
Force can require contact (two objects touching each other). Ex: Picking Up/throwing a ball (contact between hand and ball)
Some force does not require contact... gravity and magnetism act without contact only proximity (objects coming close together).
External Force ------- A force that acts on the outside of an objects material. (Outside does not necessarily mean outdoors... you can be
inside of a building... but outside of the materials). (Examples: Wind, Snow, Rain, Weight (of objects or the structure itself).
Dead Load: The external force of the WEIGHT of the Structure on itself.
Live Load: The external force of the WEIGHT of the objects and things inside or on a structure, as well as the other external
forces such as the wind, snow...etc.
Internal Force ------ A force that acts on the inside of the material of an object or structure. (Tension, Compression, Shear, Torsion)
Compression ----- A squeezing force (crushing, compacting).
Ex: Lower columns of a skyscraper are compressed by the heavy weight above them
Ex: Footprints compress the soil/ground
Ex: Garbage truck compresses the garbage to take up less space
Tension ------- A pulling force that stretches a material.
Ex: Tug-of-War with a rope
Ex: Rock Climbing (the rope in tension holding the load of a person
Ex: The cables on a suspension bridge holding up the dead load and live load.
Torsion ------- A twisting force that "turns" a material in two opposite directions.
Ex: Twisting a twizzler (liquorice)
Ex: Twisting skin on your arm (Snake-Bite)
Ex: Wringing out a wet towel
Shear ------- Bending or tearing a material due to two forces offset in opposite directions. Shear is the result of
two forces acting in opposite directions sliding past each other.
Ex: Using Scissors (sometimes called "shears"
Ex: Earth/Roadways sliding past each other and breaking due to shear force caused by an EARTHQUAKE.
Structure -------- A structure must have a definite shape/size and function (purpose).
Structures can be NATURAL (made by nature) or MANUFACTURED (man-made... influenced and made by people).
Centre of Gravity ------ A specific point where all of the mass of the structure is evenly distributed around. Gravity acts on all parts
of the structure and it will be stable if all parts are spread equally around the centre of gravity.
4 Principles of Stability: 1) Build a firm foundation
2) Balance Forces
3) Keep thrust lines vertical (Centre of gravity)
4) Use rapid rotation
Solid Structure (also called Mass Structure) -------- A structure made by the piling up (stacking) of materials
(Ex: Pillars, Snow Forts, Pyramids, Brick Walls, Mountains)
Shell Structure -------- A type of structure that obtains its strength from a thin, carefully shaped outer layer of material and
that requires no internal frame. Shell structures are strong and hollow and can support loads.
(Ex: Igloo, Egg-Shell, Cardboard Box, Garbage Bin, Wasps Nest)
Frame Structure -------- A type of structure in which a skeleton of materials supports the weight of the other parts. Frames
require different components to come together and rely on each other for support.
A joint is a point (such as a corner) where two pieces of the frame or structure are fastened together.
Stress (Structural Stress) ------- Structural stress occurs when a combination of external and internal forces act on a structure one at a
time causing changes. Stress is normal in any structure. It does not necessarily mean any changes are permanent... they may
be temporary changes. Example: When kicking a soccer ball there is a momentary compression within the material of the ball
when the foot contacts the ball.
Fatigue (Structural Fatigue) ------- Damage due to a high level of structural stress over time. Example: If a large amount of pressure is
placed on an egg there will be some compression within the shell, some tension on the surface, and these forces may lead to
cracking the shell. The cracks represent structural fatigue (damage).
Failure (Structural Failure) ------- Stress can lead to fatigue which can become failure with increased force on a structure. When a structure
FAILS it is no longer able to serve its purpose/function. It will also no longer maintain its shape, size, and form.
Structural Efficiency ------- A single number that compares the mass of a structure with the load it supports.
A high structural efficiency is a good thing.
Structural Efficiency = Maximum Mass Supported by the Structure
Mass of the Structure (Dead Load)
Foundation ------ A solid base of a structure. A firm foundation is important for stability.
Ex: The foundation of a modern home here in Winnipeg would consist of a few layers of concrete. A foundation can
prevent the heavy dead load of a structure from sinking into the ground.
Ex: Feet are generally the "foundation" or base of a human structure. The most stable position is to have both feet firmly
planted on a solid surface. Standing on tip-toes reduces the surface area on the ground and becomes an unstable foundation.
Arch ------ A strong shape often used in structural design. An arch can spread force evenly through the whole structure and down to
the ground. Ex: The Catenary Arch forms were used in the Ice Hotel Design. The arch passageways were able to evenly
distribute the weight of the snow and ice piled above it.
Corrugates ------ Corrugates or corrugation helps resist compression within a material by creating a series of arches inside of the
material. The most common example of this is corrugated CARDBOARD boxes. Corrugation increases strength and
stability of a material by using shape!
Braces, Ties, Gussets ------ A brace, tie, and gusset form a triangle in the corner of a frame or joint (where two pieces are brought
together). They change rectangular and square shapes into triangles for additional strength and stability.
Guy Wire -------- A wire used to give stability to a structure usually by counter-balancing potentially dangerous forces.
Truss -------- A frame structure that uses triangles for strength. (Ex: A Truss Bridge)
Mass -------- The mass of an object is the measure of the amount of material in it. Measurements of mass tell something about the tiniest
particles in the object.
Weight --------- Weight is similar to mass, however, it also includes the force of gravity. On the moon, you would have the same mass,
however you would weigh 1/6 less on the moon because the force of gravity is not as strong.
Gravity --------- In the 17th Century Isaac Newton realized there is a force between any two objects, anywhere in the universe, that tries to
pull them together. Newton analyzed the size of this force, which he called gravity, depended on the mass of the two objects. The
gravitational force between two very small objects is extremely small. You do not notice gravity pulling you toward trees or other
people but the force is there. Earth however, is big enough for the gravitational force between it and nearby objects to be important.
Interactions within ecosystems - glossary - part 1
Abiotic ------ refers to the nonliving factors that influence living things, such as climate, sunlight, water, geology, and minerals
Adaptations ------ features which allow a species to survive and reproduce
Atmosphere ------ "AIR" - The air layer of the biosphere (in which organisms live and travel through)
Biome ------- a large geographical region that has a similar climate condition throughout its area
(Six Biomes to know: Tundra, Boreal Coniferous Forest, Grassland, Temperate Deciduous Forest, Tropical Rain Forest, Desert)
Coniferous trees bear needles/pinecones Deciduous trees bear leaves (seasonally drop leaves that are not needed)
Biosphere ------- the layers of the earth (crust, atmosphere, and hydrosphere) that are inhabited by living things
Biotic ------ the living things within an environment (animals, plants)
Carnivores ------ animals that eat only other animals/meat for food
Cellular Respiration ---- Both plants and animals need oxygen to release energy from their food. The energy is released by a chemical change
called cellular respiration. The process takes place only inside living cells. Cellular respiration requires glucose and
oxygen. It produces carbon dioxide, water, and energy that cells use to function.
Community ------ all the populations living in one location
Climax Community ---- An ecological community in which populations of plants or animals remain stable and exist in balance with each other
and their environment. A climax community is the final stage of succession, remaining relatively unchanged until
destroyed by an event such as fire or human interference.
Competitors ------ two individuals or species that require the same resources
Consumers ------ animals which eat other organisms (plants or animals) for food (energy)
Decomposers ------ organisms that get energy from the chemical breakdown of animal or plant waste and dead organisms
(e.g. earthworms, bacteria, fungi). They break down organic material that is hard for other organisms to digest,
returning it to the environment in inorganic form to re-use by plants.
Ecology ------- the study of relationships between living organisms and their physical environment (living and non-living)
Ecosystem ------ a specific area (habitat) which organisms interact with one another and with the abiotic (non-living)
environment. An ecosystem can be a large or small area, and it must have defined boundaries.
Food Chain ------ The order of organisms through which energy is transferred. Each link (trophic level) in the chain feeds on and gets
energy from the one preceding it and in turn is eaten by the one following it, providing energy to the next level.
Food chains are seldom more than three or four links, as the energy loss at each level is so great the links cannot be
sustained beyond this.
Food Web ------ the combination of a number of food chains which show the feeding relationship between organisms.
It is food chains in an ecosystem interacting together.
Food Pyramid ------ a stylized food chain or web representing the loss of energy throughout the trophic levels
(often showing the numerical relationship)
Habitat ------ environment in which a spices lives
Herbivores ------ animals that eat plants for food (energy)
Hydrosphere ------ "WATER" the liquid water component covering about 70% of the Earth
(includes: oceans, lakes, ponds, rivers, streams...)
Lithosphere ------ "LAND" the outer part of the solid earth. Composed of rock mainly, it is the crust and upper mantle layer of the
earth.
Niche ------ the unique role of a species within an ecosystem
Omnivores ------ animals that eat both plants and other animals for food (energy)
Organism ------- any individual living thing. An organism can be made of many cells (human) or made of only one cell (micro-organisms)
Photosynthesis ------ the process by which green plants and some other organisms use sunlight to create food (sugars).
Pioneer Species ------ A pioneer species is a species that is first to establish itself in an area where nothing is growing-or in an area that
has been devastated by fire,flood, plowing etc.
Population ------ number of organisms of the same kind living in a particular area
Predators ------ carnivore that hunts for other animals (lower on the food chain) for food
Prey ------- the animal hunted by a predator
Producers ------ organisms (plants and algae) that can make their food (energy) usually through a chemical process involving sunlight
Scavenger -------- decomposer/consumers that feed on animal carcasses and garbage.
Hyenas and vultures are well-known examples but even some butterflies are scavengers. In Canada, crows are common scavengers.
Species ------- term used to describe each different kind of living thing. It is the smallest unit of classification used.
For example: Homo sapiens is the notation for humans
Succession -------- it is the progressive, gradual change of the composition of a community of organisms over time. During succession,
plant populations are constantly replaced by new species. One dominant species gives way to another.
Primary Succession ----- succession that occurs where plants have not grown before
Secondary Succession ---- succession that occurs in areas where there has been previous growth (If a natural disaster such as a forest
fire, flood, or hurricane destroy an ecosystem... the regrowth over many years is a secondary succession)
Climax Community ----- the final community in a succession
Adaptations ------ features which allow a species to survive and reproduce
Atmosphere ------ "AIR" - The air layer of the biosphere (in which organisms live and travel through)
Biome ------- a large geographical region that has a similar climate condition throughout its area
(Six Biomes to know: Tundra, Boreal Coniferous Forest, Grassland, Temperate Deciduous Forest, Tropical Rain Forest, Desert)
Coniferous trees bear needles/pinecones Deciduous trees bear leaves (seasonally drop leaves that are not needed)
Biosphere ------- the layers of the earth (crust, atmosphere, and hydrosphere) that are inhabited by living things
Biotic ------ the living things within an environment (animals, plants)
Carnivores ------ animals that eat only other animals/meat for food
Cellular Respiration ---- Both plants and animals need oxygen to release energy from their food. The energy is released by a chemical change
called cellular respiration. The process takes place only inside living cells. Cellular respiration requires glucose and
oxygen. It produces carbon dioxide, water, and energy that cells use to function.
Community ------ all the populations living in one location
Climax Community ---- An ecological community in which populations of plants or animals remain stable and exist in balance with each other
and their environment. A climax community is the final stage of succession, remaining relatively unchanged until
destroyed by an event such as fire or human interference.
Competitors ------ two individuals or species that require the same resources
Consumers ------ animals which eat other organisms (plants or animals) for food (energy)
Decomposers ------ organisms that get energy from the chemical breakdown of animal or plant waste and dead organisms
(e.g. earthworms, bacteria, fungi). They break down organic material that is hard for other organisms to digest,
returning it to the environment in inorganic form to re-use by plants.
Ecology ------- the study of relationships between living organisms and their physical environment (living and non-living)
Ecosystem ------ a specific area (habitat) which organisms interact with one another and with the abiotic (non-living)
environment. An ecosystem can be a large or small area, and it must have defined boundaries.
Food Chain ------ The order of organisms through which energy is transferred. Each link (trophic level) in the chain feeds on and gets
energy from the one preceding it and in turn is eaten by the one following it, providing energy to the next level.
Food chains are seldom more than three or four links, as the energy loss at each level is so great the links cannot be
sustained beyond this.
Food Web ------ the combination of a number of food chains which show the feeding relationship between organisms.
It is food chains in an ecosystem interacting together.
Food Pyramid ------ a stylized food chain or web representing the loss of energy throughout the trophic levels
(often showing the numerical relationship)
Habitat ------ environment in which a spices lives
Herbivores ------ animals that eat plants for food (energy)
Hydrosphere ------ "WATER" the liquid water component covering about 70% of the Earth
(includes: oceans, lakes, ponds, rivers, streams...)
Lithosphere ------ "LAND" the outer part of the solid earth. Composed of rock mainly, it is the crust and upper mantle layer of the
earth.
Niche ------ the unique role of a species within an ecosystem
Omnivores ------ animals that eat both plants and other animals for food (energy)
Organism ------- any individual living thing. An organism can be made of many cells (human) or made of only one cell (micro-organisms)
Photosynthesis ------ the process by which green plants and some other organisms use sunlight to create food (sugars).
Pioneer Species ------ A pioneer species is a species that is first to establish itself in an area where nothing is growing-or in an area that
has been devastated by fire,flood, plowing etc.
Population ------ number of organisms of the same kind living in a particular area
Predators ------ carnivore that hunts for other animals (lower on the food chain) for food
Prey ------- the animal hunted by a predator
Producers ------ organisms (plants and algae) that can make their food (energy) usually through a chemical process involving sunlight
Scavenger -------- decomposer/consumers that feed on animal carcasses and garbage.
Hyenas and vultures are well-known examples but even some butterflies are scavengers. In Canada, crows are common scavengers.
Species ------- term used to describe each different kind of living thing. It is the smallest unit of classification used.
For example: Homo sapiens is the notation for humans
Succession -------- it is the progressive, gradual change of the composition of a community of organisms over time. During succession,
plant populations are constantly replaced by new species. One dominant species gives way to another.
Primary Succession ----- succession that occurs where plants have not grown before
Secondary Succession ---- succession that occurs in areas where there has been previous growth (If a natural disaster such as a forest
fire, flood, or hurricane destroy an ecosystem... the regrowth over many years is a secondary succession)
Climax Community ----- the final community in a succession
Interactions within Ecosystems - Glossary - Part 2
Adaptations ------ Inherited traits (structural or behavioural) which allow an organism to survive in a given ecosystem or condition.
Structural Adaptation: Body parts that help an animal in a special way. For example, hollow bones make birds light, which helps them fly.
Behavioural Adaptation: Helpful instincts, or actions that the animal does automatically. Learned behaviour (such as when a dog is trained to avoid cars) is not an adaptation.
Bioaccumulation ------ The accumulation (gradual gathering) of substances transferring from the environment into an organism.
Types of substances that can bioaccumulate:
Nutrients, Toxins, Pesticides, Organic and Inorganic Compounds and Elements (such as mercury), and other pollutants.
Biomagnification ------ The tendency for substances (such as toxins) to increase in concentration as they move up the food chain.
The problem gets BIGGER! If it is a toxin or pesticide... it will affect higher level predators!
Cellular Respiration ---- Both plants and animals need oxygen to release energy from their food. The energy is released by a chemical change
called cellular respiration. The process takes place only inside living cells. Cellular respiration requires glucose and
oxygen. It produces carbon dioxide, water, and energy that cells use to function.
Cellular Respiration is how ENERGY is USED/RELEASED
Chemical Equation:
Food Energy + Oxygen --> Carbon Dioxide + Water + Energy (used)
(Sugar) (Energy for movement/growth/development)
Invasive Specie ----- An exotic (alien) non-native organism released intentionally or accidentally into a new environment. In most cases this
will change the balance of the ecosystem and impact the entire food web and balance within. 90% of invasive species
will not survive the new environment. The 10% that do make it will have a big impact!
Photosynthesis ------ the process by which green plants and some other organisms use sunlight to create food (sugars).
Photosynthesis is where ENERGY is Created and STORED
Chemical Equation:
Light Energy + Carbon Dioxide + Water (as well as nutrients) --> Oxygen + Food Energy
(Sunlight with the help of Chlorophyll)
Microscope Parts
Arm ------ holds the tube and lenses and connects to the base.
Base ------ holds up the microscope
Coarse adjustment knob ----- moves the stage up and down.
Diaphragm ---- circular disk that lets more or less light pass through
Fine adjustment knob ----- Focuses and adjusts clarity. Slightly moves the tube up and down.
High power objective lens ------ longest lens close to the slide
Light source ----- can be an actual light or a mirror that reflects light
Low power objective ------ the shortest of the lenses close to the slide
Medium power objective ------ the middle size lens close to the slide
Ocular lens (eyepiece) ----- lens closest to the eye
Revolving Nosepiece ------ allows you to rotate between the three objective lenses
Stage ----- platform for slide to rest. An opening in its centre lets light pass through.
Stage clips ----- holds the slide in place on the stage
Tube ------- gives the distance needed between the eyepiece and the objective
Structural Adaptation: Body parts that help an animal in a special way. For example, hollow bones make birds light, which helps them fly.
Behavioural Adaptation: Helpful instincts, or actions that the animal does automatically. Learned behaviour (such as when a dog is trained to avoid cars) is not an adaptation.
Bioaccumulation ------ The accumulation (gradual gathering) of substances transferring from the environment into an organism.
Types of substances that can bioaccumulate:
Nutrients, Toxins, Pesticides, Organic and Inorganic Compounds and Elements (such as mercury), and other pollutants.
Biomagnification ------ The tendency for substances (such as toxins) to increase in concentration as they move up the food chain.
The problem gets BIGGER! If it is a toxin or pesticide... it will affect higher level predators!
Cellular Respiration ---- Both plants and animals need oxygen to release energy from their food. The energy is released by a chemical change
called cellular respiration. The process takes place only inside living cells. Cellular respiration requires glucose and
oxygen. It produces carbon dioxide, water, and energy that cells use to function.
Cellular Respiration is how ENERGY is USED/RELEASED
Chemical Equation:
Food Energy + Oxygen --> Carbon Dioxide + Water + Energy (used)
(Sugar) (Energy for movement/growth/development)
Invasive Specie ----- An exotic (alien) non-native organism released intentionally or accidentally into a new environment. In most cases this
will change the balance of the ecosystem and impact the entire food web and balance within. 90% of invasive species
will not survive the new environment. The 10% that do make it will have a big impact!
Photosynthesis ------ the process by which green plants and some other organisms use sunlight to create food (sugars).
Photosynthesis is where ENERGY is Created and STORED
Chemical Equation:
Light Energy + Carbon Dioxide + Water (as well as nutrients) --> Oxygen + Food Energy
(Sunlight with the help of Chlorophyll)
Microscope Parts
Arm ------ holds the tube and lenses and connects to the base.
Base ------ holds up the microscope
Coarse adjustment knob ----- moves the stage up and down.
Diaphragm ---- circular disk that lets more or less light pass through
Fine adjustment knob ----- Focuses and adjusts clarity. Slightly moves the tube up and down.
High power objective lens ------ longest lens close to the slide
Light source ----- can be an actual light or a mirror that reflects light
Low power objective ------ the shortest of the lenses close to the slide
Medium power objective ------ the middle size lens close to the slide
Ocular lens (eyepiece) ----- lens closest to the eye
Revolving Nosepiece ------ allows you to rotate between the three objective lenses
Stage ----- platform for slide to rest. An opening in its centre lets light pass through.
Stage clips ----- holds the slide in place on the stage
Tube ------- gives the distance needed between the eyepiece and the objective